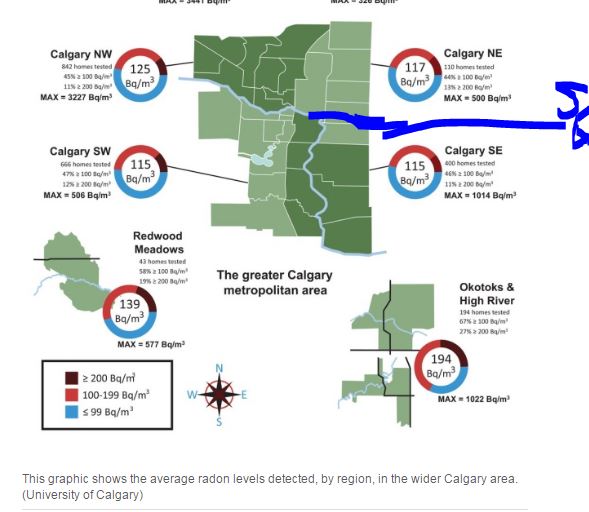
(Reuters) - The University of Oxford has launched a study to assess the safety and immune response of the COVID-19 vaccine it has developed with AstraZeneca Plc in children for the first time, it said on Saturday.
The new mid-stage trial will determine whether the vaccine is effective on people between the ages of 6 and 17, according to an emailed statement from the university.
Around 300 volunteers will be enrolled and first inoculations are expected this month, Oxford said.
The two-dose Oxford/AstraZeneca vaccine has been hailed as a “vaccine for the world” because it is cheaper and easier to distribute than some rivals.
AstraZeneca has a target to produce 3 billion doses this year and aims to produce over 200 million doses per month by April.
A HuffPost Guide To Coronavirus
- Is it safe to see people who have gotten the coronavirus vaccine?
- What it means if your partner tests positive for COVID-19 but you don’t .
- How worried should you be about the new strains of coronavirus?
- The unexpected challenges of co-parenting during a pandemic.
- 19 things we took for granted pre-pandemic but now miss .
- Find all that and more on our coronavirus hub page .
As COVID-19 cases rise, it’s more important than ever to remain connected and informed. Join the HuffPost community today. (It’s free!)
Support HuffPost
Already contributed? Log in to hide these messages.











_(720p).jpg)


 OFFICIAL HD MUSIC VIDEO.jpg)
.jpg)






































































.jpg)